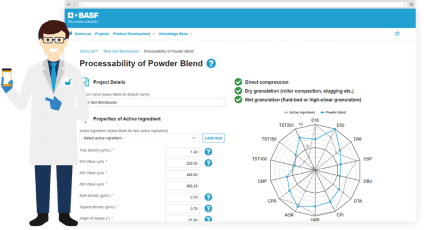
In the 1980s, BASF became the first company to apply hot melt extrusion (HME) to the production of pharmaceuticals for enhancing the solubility and bioavailability of poorly soluble actives. Today, HME is an accepted process for creating solid dispersions of APIs.
Our aim is to help our customers discover the many benefits of this technology, and assist them in leveraging it for their products. The true potential of hot-melt extrusion for pharmaceutical applications is just beginning to be uncovered – and our research continues to blaze new trails in this exciting field.
Hot Melt Extrusion with BASF Pharma Polymers
Our 2nd Edition HME Compendia offers details on polymer properties, processing, and performance advantages.